Multiscale 3D Architecture of the Salmon Cornea
- Abstract number
- 289
- Presentation Form
- Contributed Talk
- DOI
- 10.22443/rms.mmc2023.289
- Corresponding Email
- [email protected]
- Session
- FIB Applications & EM Sample Prep Techniques in Biological Sciences
- Authors
- Eran Ittah (2), Dirk Schumann (1), Natalie Reznikov (2)
- Affiliations
-
1. Fibics Incorporated
2. McGill University
- Keywords
FIB-SEM, Cornea, Collagen, Multiscale Imaging, Structural Biology
- Abstract text
Summary
FIB-SEM tomography was used, in combination with novel specimen preparation and state-of-the-art image processing tools, to obtain a high-resolution and contextualized view of the 3D architecture of the salmon cornea.
Introduction
The cornea is a unique hierarchically structured connective tissue with exceptional optical transparency, mechanical properties, and macrostructure – all of which contribute to optimal light refraction. At the macromolecular level, these properties can be attributed to the pseudocrystalline arrangement of collagen fibrils which exhibit a hexagonal packing (1). Furthermore, these arrays of collagen fibrils are arranged into lamellar structures in which two adjacent lamellae have a relatively consistent angular offset of fibrils. To maintain this unique tissue ultrastructure, these fibrillar components are decorated with highly anionic proteoglycans which enable electrostatic repulsion – exerting control over the hydration environment at the nanoscale (2). It is this hydration control which modulates the structure of the tissue and, by extension, its desirable optical properties (3).
Although the relationship between nanostructure and hydration of the cornea has been well characterized by x-ray scattering (4), this is an “averaged” representation of the bulk tissue and does not account for local microscale structural variations. Furthermore, the poor contrast of conventionally stained collagen fibrils in the cornea coupled with their relatively small diameter (30nm) limit the ability to resolve ultrastructural features with SEM over a large area. Accordingly, studies trying to elucidate the nanostructure of the cornea have been confined to 2D (TEM) or limited thickness tomographic methods (2) thus extricating the view of how this ultrastructural arrangement exists within the context of the broader tissue microstructure.
Here, we present our work in progress in the 3D analysis of corneal architecture using FIB-SEM tomography of whole Salmo salar corneas. Our aim is to enhance contrast of the corneal ultrastructure whilst maintaining pristine/hydrated fibrillar spacing by modulating osmolarity throughout the specimen preparation process. This will enable a detailed, contextualized, high resolution and multiscale 3D view of the corneal ultrastructure that has the potential to enrich the current paradigm of the hierarchical arrangement of the tissue and its relationship to hydration.
Methodology
Fresh S.salar corneas were subjected to an electron microscopy specimen preparation protocol designed to minimize the effects of post-mortem corneal swelling whilst enhancing contrast of the corneal extracellular matrix. This preparation involves the use of buffering solutions that are osmotically balanced coupled with selective staining techniques which act to both stabilize and enhance differential contrast of collagen and proteoglycans. Additionally, the protocol was accelerated employing microwave incubation which enables gentle, uniform and fast specimen processing. Finally, Imaging was performed using a Zeiss Crossbeam 550 FIB-SEM.
After FIB-SEM acquisition, 3D images were analysed using the Dragonfly software to investigate 3D fibrillar architecture. Furthermore, directionality of collagen packing was quantitatively analysed by performing 2D Fourier transform throughout the entire thickness of the cornea.
Results
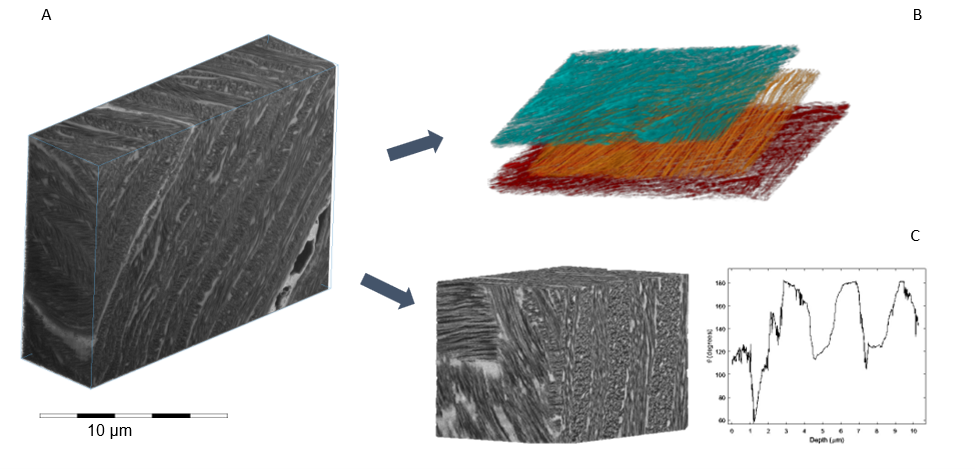
Preliminary results reveal fine 3D fibrillar architecture within a 16×13×8 µm volume of interest (A). Prior observations have asserted that the fibrils between lamellar layers exist in a nearly perfect orthogonal arrangement to one another (5). Conversely, we observe from a correlation between light and electron microscopy, that the fibrillar orientation seems to resemble rather a “St Andrew’s cross” motif existing at roughly a 60-degree offset (B,C).
Conclusion
These findings illustrate the importance of contextualization of fine features in the analysis of intricately structured tissues such as cornea. Future experiments will focus of better understanding of the architecting role of water in the organisation of corneal collagen.
- References
1. K. M. Meek, Biophys Rev. 1, 83–93 (2009).
2. P. N. Lewis et al., Structure. 18, 239–245 (2010).
3. S. Hayes et al., Journal of The Royal Society Interface. 14, 20170062 (2017).
4. K. M. Meek et al., Biophysical Journal. 60, 467–474 (1991).
5. R. D. Young et al., Experimental Eye Research. 187, 107772 (2019).