Multimodal analysis of structural & functional properties of type I collagen
- Abstract number
- 18
- Presentation Form
- Contributed Talk
- DOI
- 10.22443/rms.mmc2023.18
- Corresponding Email
- [email protected]
- Session
- Multimodal Microscopy
- Authors
- Liisa Hirvonen (1), Mina Vaez (2), Kester Ng (2), Laurent Bozec (2)
- Affiliations
-
1. The University of Western Australia
2. University of Toronto
- Keywords
FLIM, AFM, collagen
- Abstract text
Introduction
Type I collagen is one of the most abundant proteins in the human body, and is particularly important in providing mechanical resilience to connective tissue. Collagen properties can change due to ageing, disease or other damage (e.g. mechanical, chemical, radiation). Crosslinking - where collagen fibrils form strong chemical bonds with adjacent fibrils - is known to alter the physical characteristics (elasticity, thermal denaturation, morphology) of collagen structures. Crosslinking can be used to control the mechanical and biological stability of collagen in tissue engineering, where collagen is used extensively as a native structural protein to engineer scaffolds and membranes due to its biocompatibility, ease of chemical functionalization, and in vivo turnover.
In this work we characterise structural & functional changes in type I collagen in two contexts: AGE-mediated crosslinking, and after irradiation with clinically relevant X-ray doses. Glycation is a reaction related to ageing and disease, which results in stable substances called advanced glycation end-products (AGEs). AGEs accumulate within the collagen fibrils in our tissues and organs as a result of normal ageing, but they are also linked to some pathological conditions such as Alzheimer’s disease and diabetes. On the other hand, ionising radiation (e.g. X-rays, γ-rays, ultraviolet) is used routinely in clinical practice for diagnostic and therapeutic purposes, as well as a method of sterilisation for tissue implants (e.g. skin crafts), but its effects on collagen are not well characterised.
We combine multi-scale imaging and mechanical testing via Atomic Force Microscopy (AFM), Fluorescence Lifetime Imaging (FLIM) and time-lapse digital imaging to study the morphology, mechanical properties and protein stability of engineered acellular type I collagen samples that were either crosslinked with Methylglyoxal (MGO) induced AGE-crosslinking, or treated with ionising radiation at three dose levels (diagnostic, therapeutic, sterilising).
Methods
Collagen fibrils and scaffolds were produced using established protocols [1]. For AGE-mediated crosslinking experiments, the samples were incubated in 10 mM, 25 mM, 50 mM, and 100 mM MGO solutions for 3 days at 37°C for crosslink formation. For irradiation experiments, the scaffolds were dried and irradiated with three different doses (dry state analysis), or rehydrated for 24h in PBS before irradiation (wet state analysis).
An atomic force microscope (JPK Nano-wizard® 4, Bruker, Germany) was used for imaging and indentation of the collagen samples. Imaging was performed in contact mode under ambient conditions. The compressive elastic modulus was obtained by fitting a Hertzian model to the force-distance curve.
Time-correlated single-photon counting (TCSPC) FLIM was performed with a confocal laser scanning microscope (A1R, Nikon, Japan) with a 20x NA0.75 objective and a FLIM add-on module (LSM Upgrade Kit, PicoQuant, Germany). Fluorescence was excited with a picosecond pulsed 405 nm diode laser operating at 5 MHz repetition rate, and the average lifetime was calculated from a three-exponential function fitted to the sum decay.
Results
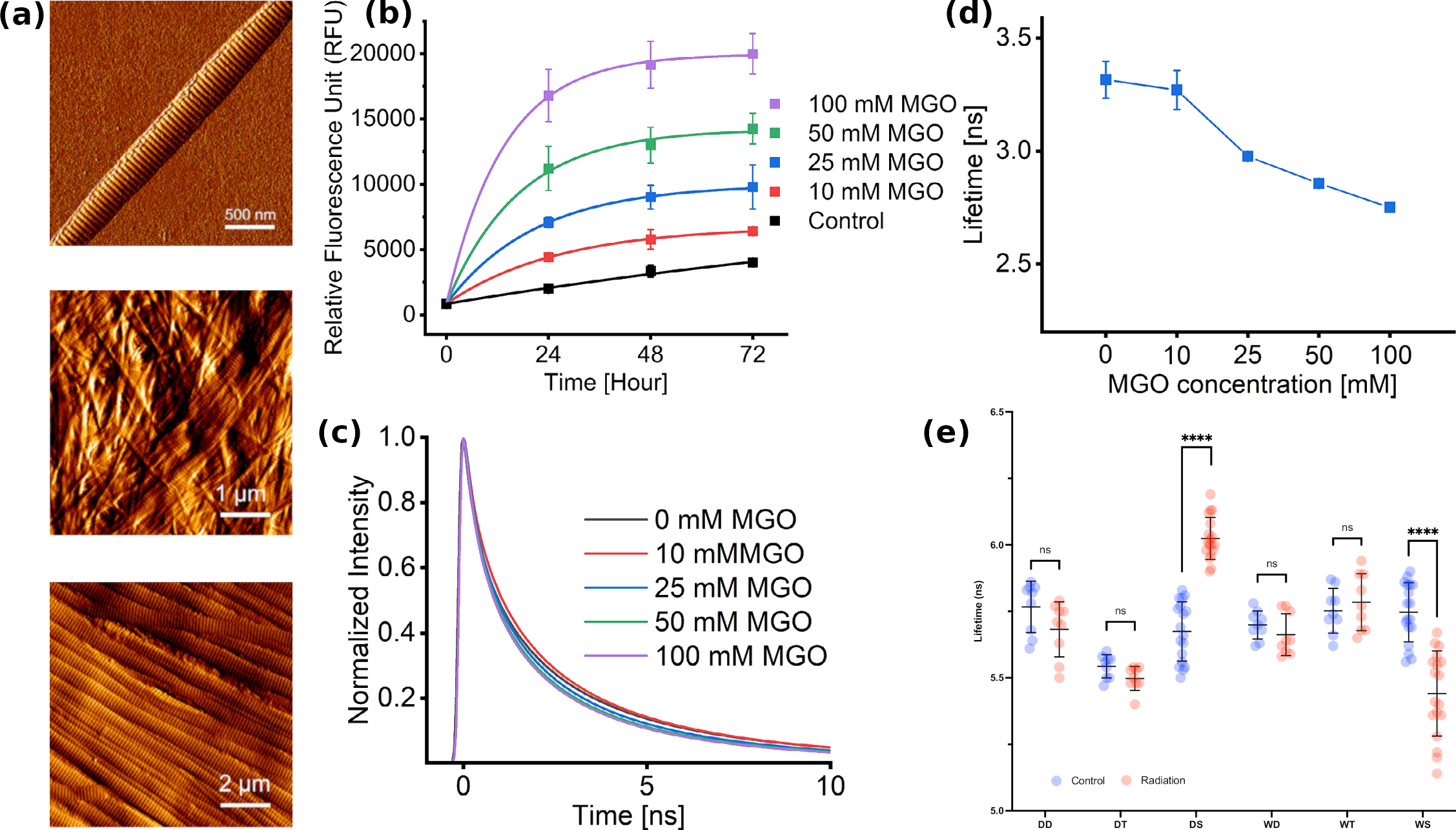
Fig 1a shows representative images of a single collagen fibril, collagen gel with random fibril orientation, and oriented collagen fibrils in rat tail tendon. No morphological changes were detected in the AGE-crosslinked samples at the fibril level, but more fibres were seen with increasing MGO concentration.[2] Mechanical indentation measurements show increased scaffold stiffness, but no change in the individual fibril stiffness. This is consistent with the formation of crosslinks that bundle fibres together without modifying the collagen structure. No morphological changes were detected in the irradiated samples.
In the AGE-crosslinked samples, autofluorescence was measured during crosslinking and was found to increase during crosslink formation (Fig 1a). The glycation reaction was stopped after 3 days, and fluorescence lifetimes were measured. Fig 1b shows representative decays which show faster decays for higher MGO concentrations; the calculated lifetimes also show decrease (Fig 1c). Crosslinks are often fluorescent, so increase in intensity is expected. However, since the fluorescence lifetime is not influenced by fluorophore concentration, the shortening of the lifetime of different glycated scaffolds can be explained through the formation of different fluorescent species or AGEs in our case. This decrease in lifetime values demonstrates that new crosslink species formation is dependent on the MGO concentration.
In the irradiated samples, changes in mechanical properties were detected in different directions (stiffening with wet irradiation, softening with dry irradiation). Fluorescence lifetime was also found to change in different directions as a function of hydration. This supports the hypothesis of alternate mechanisms by which ionising radiation interactions with the collagen fibrils mediated by water.
Conclusions
We identify unique variations in the properties of collagen following in vitro tissue glycation by MGO and propose this method of collagen crosslinking as a means to modulate the biophysical properties of collagen fibrils and scaffolds prior to cell seeding or clinical implantation.
We also demonstrate that ionising radiation can alter the mechanical properties of collagen fibrils with minimal morphological change and that these changes arise from inter-fibrillar changes. Clinically, these changes may have implications in the pathophysiology and management of radiation-related sequelae, including osteoradionecrosis and cancer dissemination.
Fig 1: (a) Representative topological AFM images of (top) single collagen fibril, (middle) collagen fibrils in scaffold, (bottom) extracted collagen fibrils in rat tail tendon, (b) Autofluorescence of glycated collagen scaffolds over a 3-day incubation period, (c) Representative fluorescence decays of glycated collagen scaffolds, (d) Mean fluorescence lifetimes of collagen scaffolds, (f) Mean fluorescence lifetimes of irradiated collagen scaffolds.
- References
[1] Brown RA, Wiseman M, Chuo CB, Cheema U, Nazhat SN. “Ultrarapid Engineering of Biomimetic Materials and Tissues: Fabrication of Nano- and Microstructures by Plastic Compression”. Adv Funct Mater. 15(11):1762-70, 2005.
[2] M. Vaez, M. Asgari, L. Hirvonen, G. Bakir, E. Khattignavong, M. Ezzo, S. Aguayo, C. M. Schuh, K. Gough, L. Bozec. “Modulation of the biophysical and biochemical properties of collagen by glycation for tissue engineering applications”. Acta Biomaterialia, 155:182-198, 2023.