Atomic Imaging and Spectroscopy of Grain Boundaries, Crystallisation and Heterostructure Interface Characteristics Within Solid-State Battery Materials
- Abstract number
- 216
- Presentation Form
- Contributed Talk
- Corresponding Email
- [email protected]
- Session
- Sustainability and Carbon Net Zero
- Authors
- Mr Connor Murrill (1), Prof Vlado Lazarov (1), Dr Richard Douthwaite (1), Dr Demie Kepaptsoglou (1)
- Affiliations
-
1. University of York
- Keywords
Solid-state
Batteries
High-resolution
Lithium
STEM
TEM
Spectroscopy
Heterostructure
Grains
Boundaries
Symmetry
- Abstract text
The work presented consists of three distinct stages of analysis. Grain boundaries in LiCoO2 (LCO) electrode films, deposited at low temperatures, and the consequences of nanograin orientation. Secondly, crystallisation characteristics of Li7La3Zr2O12 (LLZO) electrolyte films have been compared after using different pulsed laser deposition (PLD) and annealing temperatures. Finally, heterostructure samples on SrTiO3 (STO) and gadolinium gallium garnet (GGG) substrates have provided a basis for detailed analysis of temperature effects during or after bilayer deposition. Atomic structures and chemical mixing, analysed by electron energy loss spectroscopy (EELS), at the interfaces have revealed thermal decomposition characteristics.
Introduction
Solid-state electrodes and electrolytes provide a potential path to improving the capacity and performance characteristics of lithium-ion energy storage devices.[1, 2] Optimising these systems provides several challenges, including optimising the crystalline characteristics within micron and nanoscale grain networks, alongside electrochemical interface decomposition.[1, 3, 4] The electrode and electrolyte focused upon in this study are LiCoO2 and Li7La3Zr2O12. With simple electrode (LCO) chemistry and structure to model from high resolution electron microscopy and LLZO being identified potential solid-state electrolyte with high lithium conductivity, both are relevant materials for research into progressing scientific literature.[5, 6]
With resolutions achievable below 1 Å in aberration corrected transmission electron microscopy (TEM), structural analysis of grains, grain boundaries, as well as interfaces between LCO and LLZO provides exciting opportunities. Observing grain alignment with respect to specific misorientations, symmetry or defects within their bulk, helps understand the limitations to lithium conductivity different preparation methods may provide. Alongside imaging, chemical mapping using high-resolution EELS, at interfaces between two materials can inform crystal modelling and calculations of how these influence lithium transfer over boundaries within the bulk of each material and across their interface.
Methods
Thin films of LCO and LLZO have been deposited using pulsed laser deposition (PLD) at a range of temperatures. LCO deposition focused on low-temperature deposition onto Al2O3 (ALO) and SrTiO3 (STO) substrates to understand the orientation, lithium deficiency and grain dynamics.
LLZO was deposited onto STO and GGG substrates with a focus on crystallisation in-situ at 600oC during PLD and ex-situ (furnace annealing) after low temperature, amorphous films were acquired by PLD.
Heterostructures were created by deposition LCO onto STO and then LLZO atop the LCO layer. Annealing was conducted at 600oC (4 hours) to try and crystallise the LLZO layer. This was reduced to 30 minutes due to a damaged LLZO layer and lithium deficiency during prolonged annealing. On GGG samples followed the layering order GGG, LLZO and LCO, given LLZO has a close lattice match with GGG and orientated crystalline growth was expected from work done on single layer samples.
A focused ion beam has been used to create the electron transparent lamella for TEM and STEM. SEM has been used to observe the surface morphologies of the thin films.
Grain Boundary Symmetries in LiCoO2
The atomic-scale analysis of LCO films, deposited at low-temperatures (<200oC) on ALO, with a (0001) surface orientation, displayed three phases. The low-temperature cubic, high-temperature rhombohedral and lithium deficient Co3O4 phases (Figure 1, a).[7] Rhombohedral grains showing distinct cobalt oxide layering either formed twinned boundaries with an anti-phase alignment between cobalt layers for the [100] neighbouring the [-100] lattice orientation (Figure 1, a). Although the misorientated rhombohedral grains seen in Figure 1, b can form in phase layers if in contact at a boundary. The cubic grains, viewed along the [110] direction led to a well-aligned interface with the rhombohedral grains along a [100] axis in the same direction, any twinned grain (e.g. rhombohedral [-100]) will form a mismatched interface with the cubic grain. The same applied to the spinel phase symmetry, showing nanoscale grain orientations play an important role in alignment of lithium migration channels.
Figure 1
Crystallisation of LLZO Electrolyte Thin Films
Samples crystallised from amorphously deposited films (PLD below 600oC) at temperatures exceeding 900oC, formed polycrystalline nanograins, with no relationship between substrate orientation (Figure 2, a). These films appeared to have significant lithium deficiency. In contrast, deposition with the substrate held above 600oC during PLD achieved crystalline deposition with some substrate orientation dependence on STO (Figure 2, b) and GGG. Image and diffraction pattern analysis suggests partial lithium deficiency, especially given large grains of La2Zr2O7 (LZO) along the [100] axis can be seen in LLZO films grown on STO(111) (top right corner of Figure 2, b).
Figure 2
LCO/LLZO Heterostructures
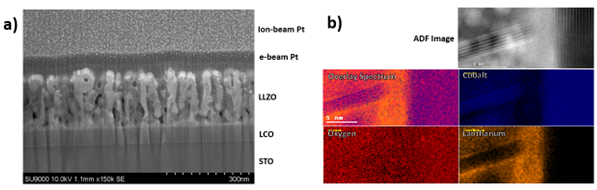
Initially heating the sample to 600oC for 4 hours produced a porous electrolyte with significant damage either from the heating process or gallium ion beam (Figure 3, a). Later samples suggest this was temperature related damage owing to the lack of similar problems with samples annealed at 600oC for 30 minutes. From images and EELS data collected at the interface it is apparent, significant La and Co intermixing can occur. Small crystalline regions returned maps, as shown in Figure 3, with compositions of lanthanum-cobalt-oxide or growth of what appeared to be crystallites of Li1-xCoO2 penetrating around 20 nm through the interface (Figure 3, b). Annealing also created strongly orientated growth along the (001) plane for LCO on STO(111) and (104) on STO/(100), compared to the three phase system seen in low temperature LCO films in Figure 1.
Figure 3
Conclusions
Three distinct areas of focus have uncovered atomically resolved grain boundaries within LCO and LLZO thin films. Analysis of LCO has revealed the dependence on orientation of adjacent grains with respect to the oxygen and cobalt symmetry. Three key phases exist, all interacting with each other with consistent lattice orientation relationships leading to twinned, well-aligned and/or anti-phase boundaries.
Amorphous LLZO crystallises out into polycrystalline, nanogranular films with significant lithium deficiency at temperatures above 900oC, or 600oC with prolonged annealing times. Temperatures of 600oC during PLD lead to crystalline materials with orientations dependant on the STO crystallinity.
Heterostructure interfaces between the two materials appear to decompose into a mixture of lanthanum oxide, lanthanum cobalt oxide, with some penetration of crystalline LCO. Further investigation into these interfaces is ongoing to support these initial findings.
- References
1. Yu, X. and A. Manthiram, Electrode–electrolyte interfaces in lithium-based batteries. Energy & Environmental Science, 2018. 11(3): p. 527-543.
2. Bachman, J.C., et al., Inorganic Solid-State Electrolytes for Lithium Batteries: Mechanisms and Properties Governing Ion Conduction. Chemical Reviews, 2016. 116(1): p. 140-162.
3. Mariappan, C.R., et al., Grain boundary resistance of fast lithium ion conductors: Comparison between a lithium-ion conductive Li–Al–Ti–P–O-type glass ceramic and a Li1.5Al0.5Ge1.5P3O12 ceramic. Electrochemistry Communications, 2012. 14(1): p. 25-28.
4. Dawson, J.A., et al., Atomic-Scale Influence of Grain Boundaries on Li-Ion Conduction in Solid Electrolytes for All-Solid-State Batteries. Journal of the American Chemical Society, 2018. 140(1): p. 362-368.
5. Li, Z., et al., Structural study of epitaxial LiCoO2 films grown by pulsed laser deposition on single crystal SrTiO3 substrates. Thin Solid Films, 2016. 612: p. 472-482.
6. Sastre, J., et al., Lithium Garnet Li7La3Zr2O12 Electrolyte for All-Solid-State Batteries: Closing the Gap between Bulk and Thin Film Li-Ion Conductivities. Advanced Materials Interfaces, 2020. 7(17): p. 2000425.
7. Porthault, H., et al., Raman study of the spinel-to-layered phase transformation in sol–gel LiCoO2 cathode powders as a function of the post-annealing temperature. Vibrational Spectroscopy, 2012. 62: p. 152-158.