Advanced Life Science SEM-EDS analysis using an annular silicon drift detector
- Abstract number
- 125
- Presentation Form
- Poster
- DOI
- 10.22443/rms.mmc2023.125
- Corresponding Email
- [email protected]
- Session
- Poster Session Three
- Authors
- Max Patzschke (1), Dr Menzies Andrew (1)
- Affiliations
-
1. Bruker Nano
- Keywords
life science, EDS, SEM, annular detector, mineralization, overcome charging effects, avoid shadow effects, topographic samples, uncoated samples, excluded samole preperationsting nettle plant, plants, FlatQUAD, fast mapping, low bream currents, low kV analysis
- Abstract text
SEM-EDS is often challenging when it comes to life science samples and elemental analysis. Especially when standard sample preparation methods, like carbon-coating or polishing, are not possible. Then on topographic complex samples, data shadow effects and charging occurs for uncoated samples. In addition, low beam currents often must be applied so that the sample is not damaged, and this subsequently leads to low count rates and potentially long analytical times.
To overcome such analytical challenges, an annular detector (Figure 1) was used for the study on life science samples. This detector is inserted between the pole-piece and the sample and is ideally suited for the analysis of topographically complex and beam sensitive samples. Shadowing effects are minimized as there are four separate X-ray detector segments that cover a large solid angle of 1.1sr. This allows sufficient data to be collected at low beam currents, where a sample can be investigated without carbon coating under high vacuum. Compared to low vacuum analysis, this results in higher spatial resolution of the SEM images and X-ray element mappings.
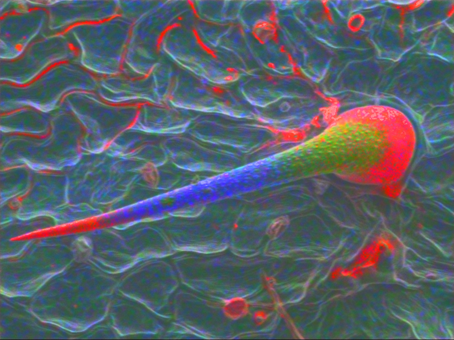
A sting nettle plant (Urtica sp. (Figure 2)) was analyzed at 6kV using low beam currents to avoid charging and beam damage on the sample.
The tip and base of the trichome have Si-rich minerals which acts as a hardening agent in the material but may also break easily when necessary. The rest is made of Ca- and Mg-rich minerals, giving the structure the necessary flexibility.
Studying the elemental composition and distribution in mineralized tissues considerably helps to understand plant anatomy, physiology and ecology
Here we will present examples from a life science study of the mineralization processes in sting nettle plants, sea urchin, a bacterium and microbial mat of diatoms.
Figure 1: Annular SDD detector which is placed like a BSE detector between pole piece and sample to cover a large solid angle of 1.1sr.
Figure 2: Sting of a nettle plant with different gradients in the stage of bio mineralization Si (red) Ca (blue) and Mg (green).