Analysis of three-dimensional cellular scaffolds using multimodal imaging
- Abstract number
- 187
- Presentation Form
- Poster
- DOI
- 10.22443/rms.mmc2023.187
- Corresponding Email
- [email protected]
- Session
- Poster Session Two
- Authors
- Miss Camilla Dondi (1, 3, 2), Dr Dimitrios Tsikritsis (1), Professor Bart Hoogenboom (2, 3), Dr Max Ryadnov (1), Dr Michael Shaw (1)
- Affiliations
-
1. National Physical Laboratory
2. University College London
3. London Centre for Nanotechnology
- Keywords
Multimodal imaging, 3D scaffolds, hydrogels, AFM, light microscopy, SRS, SHG
- Abstract text
Scaffolds for 3D cell and tissue culture are widely used as platforms for regenerative medicine and drug screening. However, much remains unknown about how interactions between cells and scaffolds influence cell survival and proliferation and how these cell responses can be controlled through rational scaffold design. The main aim of this study is to apply multimodal microscopic imaging to investigate the structural, mechanical and chemical characteristics of cell-seeded scaffold systems and establish how these properties affect cell behaviour.
A range of hydrogels (3D polymer networks) have been explored as preferred scaffold solutions for in vitro disease models, nanocarriers for drug delivery or vaccines. Scaffolds vary significantly in their material composition, degradability, pore size and stiffness. Many systems have proven to be biocompatible and able to support cell encapsulation and attachment. Nonetheless, it remains unclear how these matrices instruct cell development and fate. Spatial mapping of the structural, chemical and functional properties of the scaffold and the cells cultured within is therefore crucial in order develop improved understanding of cell-matrix interactions. State-of-the-art analytical techniques now make it possible to characterise and correlate the properties of biomaterials, to support the development of more reliable and consistent 3D scaffolds.
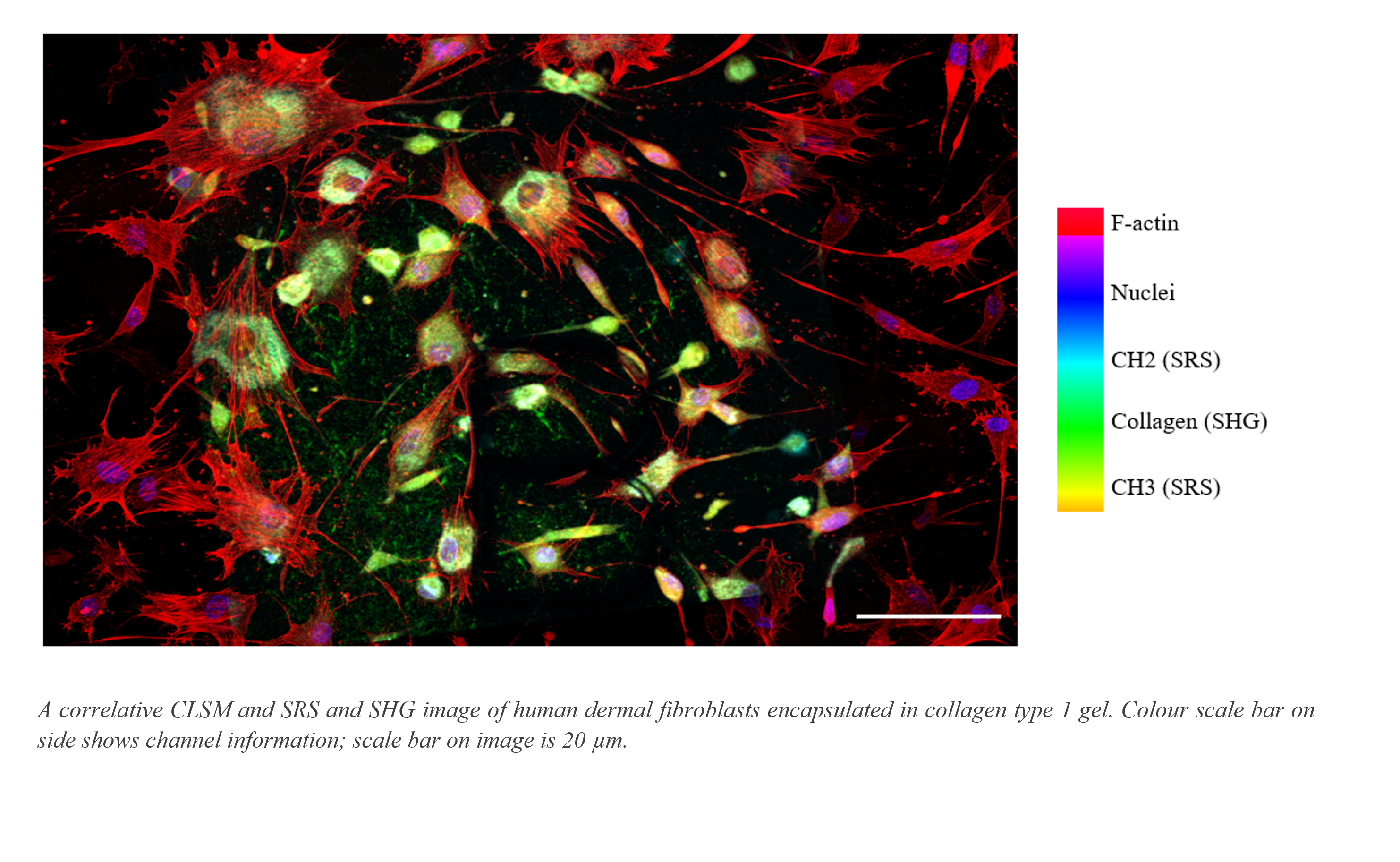
In this study we developed a multimodal imaging pipeline to exploit information accessible via different techniques to investigate the physicochemical properties of two hydrogels: a collagen type 1 scaffold derived from rat tail and a novel synthetic system. Multimode AFM in peak force tapping mode allowed topographical analysis and quantification of the length and thickness of matrix fibres, factors which can influence the mechanical properties of the associated hydrogels. F-actin visible in confocal laser scanning image data (see image), allowed assessment of human dermal fibroblast cellular morphology, with bundling of F-actin in the cytoskeleton indicating the formation of filopodia through which cells probe and interact with the matrix. The chemical information accessible in through SRS enabled measurement of CH2 and CH3 content in the cell membrane and nuclei (see image), whilst SHG signals allowed direct label-free visualisation of collagen fibrils.
These findings suggest a set up for a multimodal analysis as a solution to characterise cell-seeded scaffolds systems. The combination of different imaging modalities captures comprehensive structural and chemical information. This is essential to support the quality control of existing cell scaffolds and the rational design of new scaffold systems.